Volume 23, Issue 3 (September 2025)
Iranian Rehabilitation Journal 2025, 23(3): 331-338 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Hussein Z A, Khazraji R T T, Gazi H K. The Efficacy of Strengthening and Stretching by PNF for Foot Drop in Hemiplegia. Iranian Rehabilitation Journal 2025; 23 (3) :331-338
URL: http://irj.uswr.ac.ir/article-1-2489-en.html
URL: http://irj.uswr.ac.ir/article-1-2489-en.html
1- Department of Physiotherapy Techniques, College of Health and Medical Technologies, Middle Technical University, Baghdad, Iraq.
Keywords: Hemiplegia, Foot drop, Proprioceptive neuromuscular facilitation (PNF), Stroke rehabilitation
Full-Text [PDF 785 kb]
(1587 Downloads)
| Abstract (HTML) (2323 Views)
Full-Text: (448 Views)
Introduction
Foot drop is a prevalent gait impairment in hemiplegia, resulting from disruption of corticospinal pathways due to upper motor neuron lesions, which causes spastic paresis and weakness of the tibialis anterior (TA) muscle [1]. This leads to insufficient ankle dorsiflexion during the swing phase, resulting in a compensatory steppage gait. Hemiplegic foot drop is further exacerbated by the co-contraction of plantar flexors (e.g. gastrocnemius-soleus complex), which contributes to a limited range of motion (ROM) and fall risk [2].
Proprioceptive neuromuscular facilitation (PNF) incorporates proprioceptive input to stimulate cortical reorganization and motor learning [3]. PNF is a neurophysiological intervention that enhances neuromuscular responses through targeted stimulation of proprioceptors, including muscle spindles and Golgi tendon organs [4]. By modulating proprioceptive feedback, PNF augments sensorimotor integration, thereby improving muscle strength, flexibility, balance, and intermuscular coordination [3]. This technique effectively recruits maximal motor unit activation, critical for functional rehabilitation. The technique leverages diagonal synergistic movement patterns (e.g. D1/D2 flexion-extension sequences) and isotonic/isometric contractions to strengthen weakened muscles, while also employing force irradiation, a process in which neural impulses propagate through spinal interneuronal pathways to enhance contractions in synergistic muscle groups. This technique utilizes diagonal movement patterns, specifically D1 (hip flexion-adduction-external rotation, ankle dorsiflexion-inversion) and D2 (hip flexion-abduction-internal rotation, ankle dorsiflexion-eversion), to improve neuromuscular control [3, 5, 6]. These patterns utilize Sherrington’s principles of irradiation and successive induction, whereby resistance imposed on more powerful synergists (e.g. hip flexors) facilitates the recruitment of less powerful muscles (e.g. TA) through spinal interneuronal facilitation [3-6].
Prior investigations into PNF for foot drop have demonstrated improvements in ROM, muscle strength, and gait parameters, particularly among stroke survivors [7, 8]. For instance, Kim et al. highlighted enhanced balance and gait efficiency in hemiplegic patients undergoing PNF combined with functional electrical stimulation [7]. At the same time, Pasiut et al. observed synergistic benefits of PNF and botulinum toxin in the management of spasticity [8]. However, these studies often focus on short-term outcomes (e.g. 4–6 weeks). This study addressed this limitation by evaluating a 14-week PNF intervention incorporating D1/D2 patterns and force irradiation.
Materials and Methods
Setting and study design
This study employed a single-arm, pre-post intervention design to assess the effectiveness of PNF techniques in individuals with hemiplegia and foot drop. The study included patients who attended two clinical sites in Baghdad: The Medical Baghdad Teaching Hospital and the Sadr Al-Qanat Center (Medical Rehabilitation Center and Joint Diseases), from February 15, 2023, to August 31, 2023.
Eligibility criteria
The inclusion criteria included adults (21–60 years) with spastic hemiplegia following stroke. They displayed foot drop (defined as diminished dorsiflexion of the ankle during the swing phase of gait) [7].
The exclusion criteria included flaccid hemiplegia, cerebral palsy, lower motor neuron disorders, psychosomatic conditions, uncontrolled cardiovascular or respiratory disorders, and inability to cooperate with the therapy protocol.
Intervention
The PNF protocol was conducted twice a week for 14 weeks, and each session lasted 60 minutes, consisting of 10–15 minutes of moderate-intensity aerobic exercise (cycloergometric training). After a 6-second isometric contraction (contract-relax PNF), PNF stretching targeting the TA, extensor hallucis longus, extensor digitorum longus, and peroneus tertius, two sets of 30-second holds (respective muscle group targeted) were performed. Resistance exercises were gradually added, which comprised manual resistance in dorsiflexion (three sets of 10 repetitions) and indirect resistance through knee flexion to facilitate dorsiflexion (two sets of 15 repetitions). D1 and D2 PNF patterns involving hip flexion/adduction, ankle dorsiflexion, and toe extension were also included in the intervention. Resistance intensity or the number of repetitions was adjusted for each individual every 15 days, according to tolerance [5] (Figure 1).

Outcomes
This study examined the results in two stages: Before the intervention and after a 14-week intervention.
Muscle strength: Muscle strength (0–5) was assessed using manual muscle testing according to the Medical Research Council (MRC) scale. The strength of the quadriceps, hamstrings, TA, and calf muscles was investigated. For the quadriceps, the participant lay supine with the knee in extension. Resistance was applied to the distal anterior thigh while the participant attempted to extend the knee. The hamstring strength test was performed in the prone position by applying resistance to the posterior heel while maintaining knee flexion at 90°. The TA strength was measured in the supine position with the knee slightly flexed at 90°, resisting the dorsiflexion force applied to the dorsal foot. Calf muscle (gastrocnemius/soleus) strength was assessed in a prone position with the knee extended, and resistance was applied to the plantar aspect of the heel during plantar flexion. Each muscle group was graded according to the MRC scale: 0 (no contraction), 1 (flicker of contraction), 2 (movement in the absence of gravity), 3 (movement against gravity), 4 (movement against minimal resistance), and 5 (normal strength) [9-11].
Spasticity: The modified ashworth scale (MAS) was employed to examine the resistance to passive movement in the quadriceps, hamstrings, TA, and calf muscles as an assessment of spasticity. For the quadriceps, the participant lay supine with the knee extended, and the examiner passed the knee through available flexion while assessing resistance encountered during the arc of movement. For hamstring spasticity assessment, participants were positioned prone with their knees flexed at 90°, and resistance was assessed during passive knee extension. TA spasticity was evaluated in the supine position by passive ankle plantar flexion and inversion, and spasticity of the calf muscle (gastrocnemius/soleus) was evaluated in the prone position with the knee extended, providing resistance to passive ankle dorsiflexion. Each muscle group was assessed using the MAS (0, no increase in tone; 1, slight increase in tone with minimal resistance at the end-range; 2, moderate increase in tone with resistance on most of the range; 3, severe increase in tone with stiff movement; and 4, rigid fixation) [12, 13].
Function: The outcome was evaluated using the forward reach test (FRT). The participants stood barefoot on a level surface, with their feet shoulder-width apart and their arms at 90° of shoulder flexion (parallel to the floor). We placed a measuring tape in a fixed vertical position at the level of the participant’s third metacarpal, aligned with the participant’s acromion process. The participants were asked to reach as far as possible, keeping their feet and trunk aligned and standing with their assistive devices in place. The maximum gross distance (cm) reached in three consecutive attempts was measured, and the mean value was used for analysis [14, 15].
Ankle dorsiflexion ROM: The passive ankle dorsiflexion ROM was quantified in each participant by measuring angular displacement with an electrogoniometer. The object was placed in the supine position with the knee fully extended to avoid tension in the gastrocnemius-soleus complex. The lateral malleolus served as the axis for the electro goniometer, with one arm positioned along the fibular shaft (proximal segment) and the other positioned along the lateral aspect of the foot (distal segment). The examiner used slow, controlled force to passively bring the ankle into maximal dorsiflexion until resistance was felt or the end range was reached. The peak dorsiflexion angle (in degrees) was obtained during three consecutive trials, and the mean value was utilized for data analysis [16].
Statistical analysis
A priori power analysis using G*Power software, version 3.1 determined a required sample size of 30 participants for the Wilcoxon signed-rank test, with parameters set at a medium-to-large effect size (r=0.5) for MAS [8], α=0.05 (two-tailed), and power (1-β)=0.80, ensuring sufficient statistical power. The Wilcoxon signed-rank test was then utilized to compare pre- and post-intervention scores for muscle strength (MRC scale) and spasticity (Ashworth scale), given their non-normal distribution and ordinal nature; effect sizes were quantified using Pearson’s correlation coefficient (r≥0.5 indicating large effects). For continuous variables, normality was confirmed for FRT scores and ankle dorsiflexion ROM via histograms and Shapiro-Wilk tests (P>0.05), allowing for the application of paired t-tests; effect sizes were determined using Cohen’s d (d≥0.8 denoting large effects). A significance level of P<0.05 (two-tailed) was used for all statistical tests.
Results
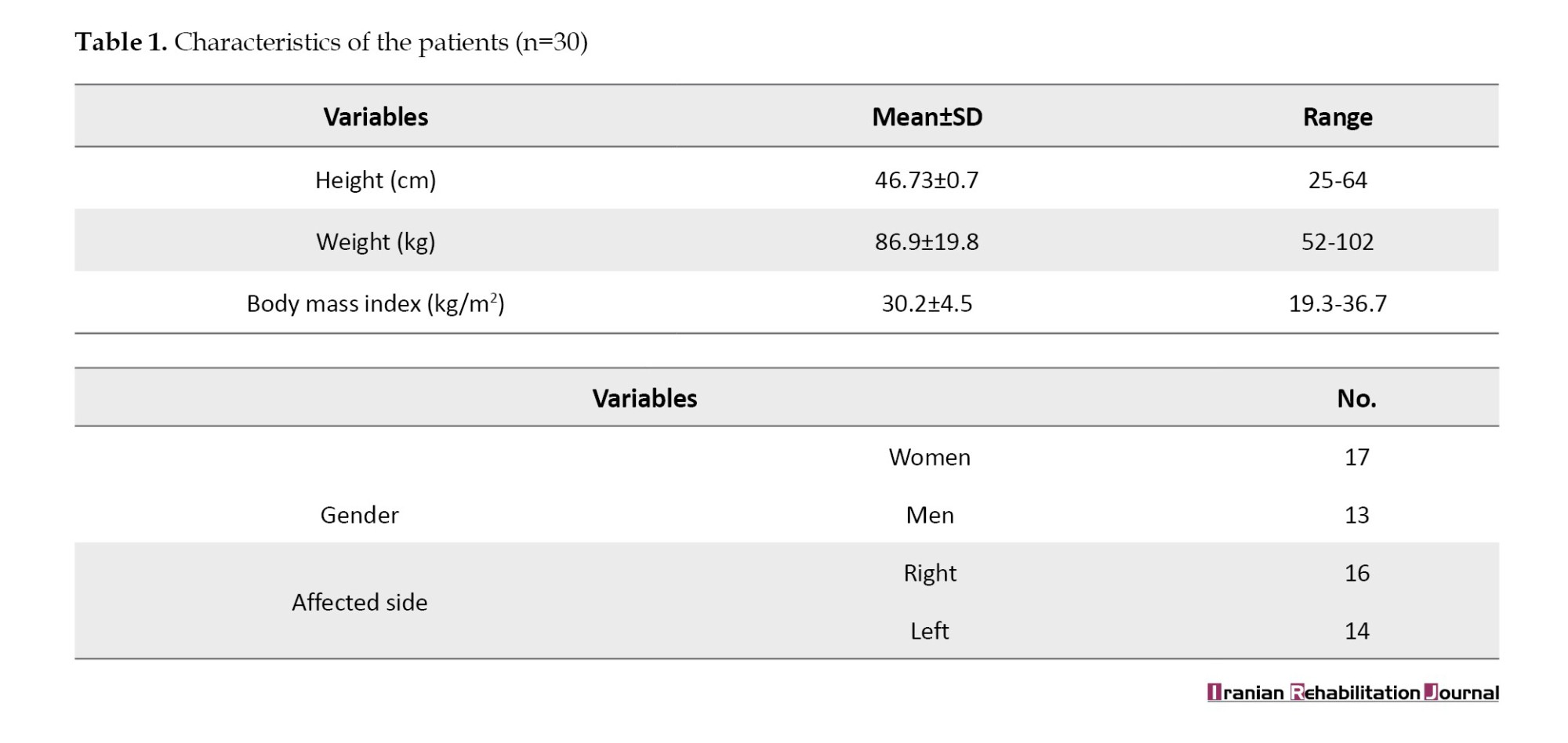
Thirty participants completed the study per protocol, with no withdrawals or protocol deviations. Table 1 presents baseline characteristics. The intervention demonstrated excellent safety, with zero serious adverse events (e.g. musculoskeletal injuries, cardiovascular complications) reported during the 14-week trial.
Muscle strength outcomes, as measured by the MRC scale, showed significant improvement post-14-week PNF intervention. The TA showed a median increase in grade from 2 (pre-intervention) to 3 post-intervention, reflecting a gain in full ROM against gravity to moderate resistance. Before therapy, 92% of subjects were at grade 2 (complete range without gravity), whereas, after therapy, they were 52% grade 3 (complete range against gravity), and 42% grade 4 (moderate resistance). The median quadriceps strength increased significantly from 2 to 4 (P<0.01), and 63% achieved quadriceps strength ≥ grade 4 (vs 28% pre-intervention, P=0.007). Hamstring and calf muscle grades also improved, with median grades increasing from 2 to 3 for hamstrings (58% at grade 3+ post-intervention compared to 12% pre-intervention) and from 2 to 4 for calf muscles (60% at grade 4 + post-intervention vs 20% pre-intervention). These changes were statistically significant (Table 2).
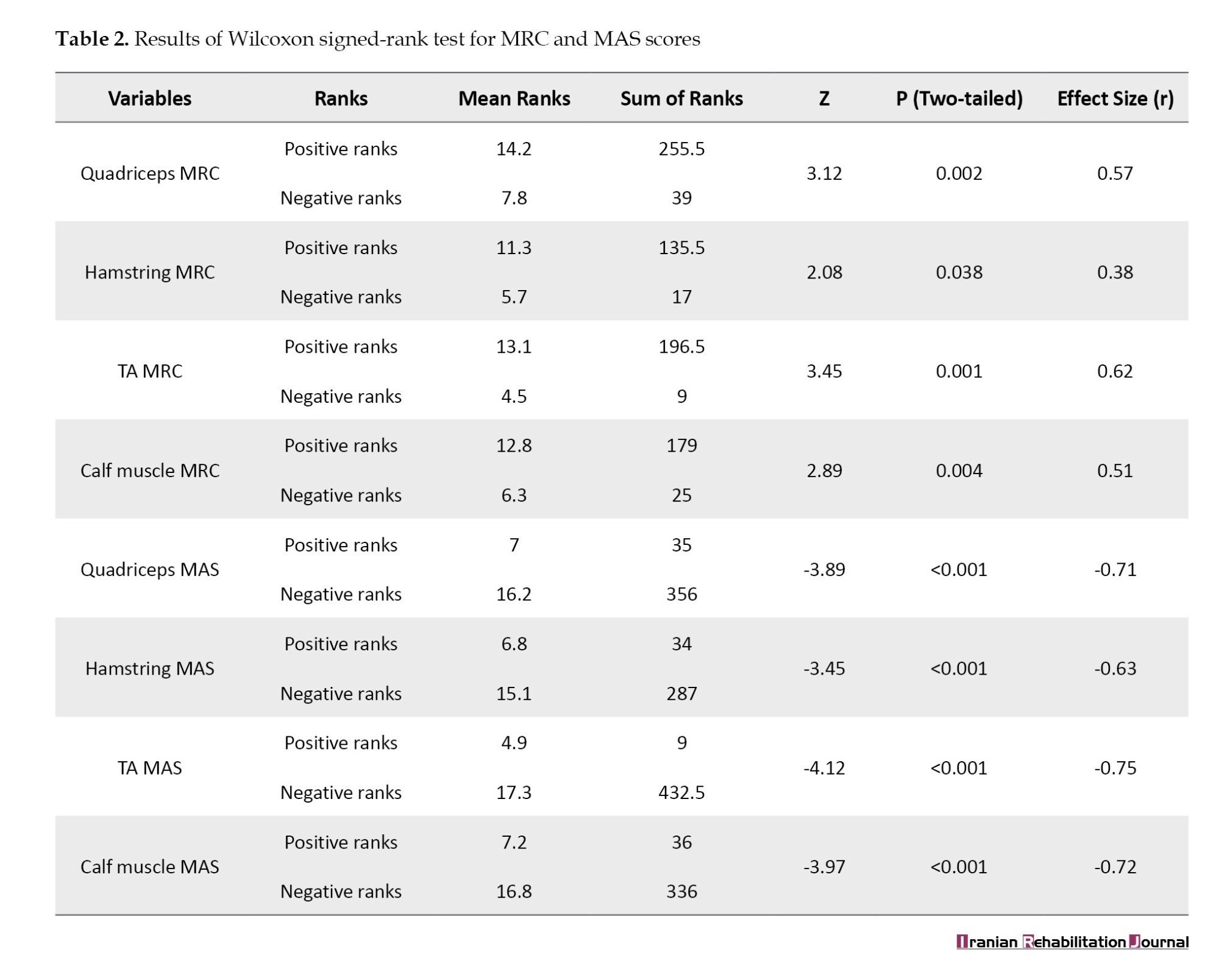
The 14-week PNF intervention led to significant reductions in spasticity across all assessed muscles (P<0.001). TA spasticity decreased from a median MAS grade of 3 (pre-intervention) to 2 post-interventions, reflecting a shift from marked rigidity to moderate resistance. Pre-intervention, 78% of participants exhibited grade 3–4 spasticity, which reduced to 32% post-treatment, with 54% achieving grade 2 and 14% grade 1. Quadriceps spasticity improved from a median grade of 3 to 2, with 65% of participants achieving a grade 2 or lower (compared to 18% pre-intervention). The hamstrings and calf muscles also showed substantial reductions: The hamstrings shifted from a median grade 3 to 2 (60% at grade 2 post vs 12% pre), and the calf muscles decreased from a median grade 3 to 1 (48% at grade 1 post vs 8% pre). These changes were statistically robust (P<0.001), with large effect sizes (r=0.63–0.75) (Table 2).
Statistical analysis confirmed that the mean FRT scores at 14 weeks improved significantly from baseline (Table 3). The mean FRT score was 26.43±2.84 cm before the intervention and 31.64±1.63 cm after the intervention. The mean difference of 5.21 cm between phases had a significant difference (P<0.001) with an associated large effect size (Cohen’s d=0.83).
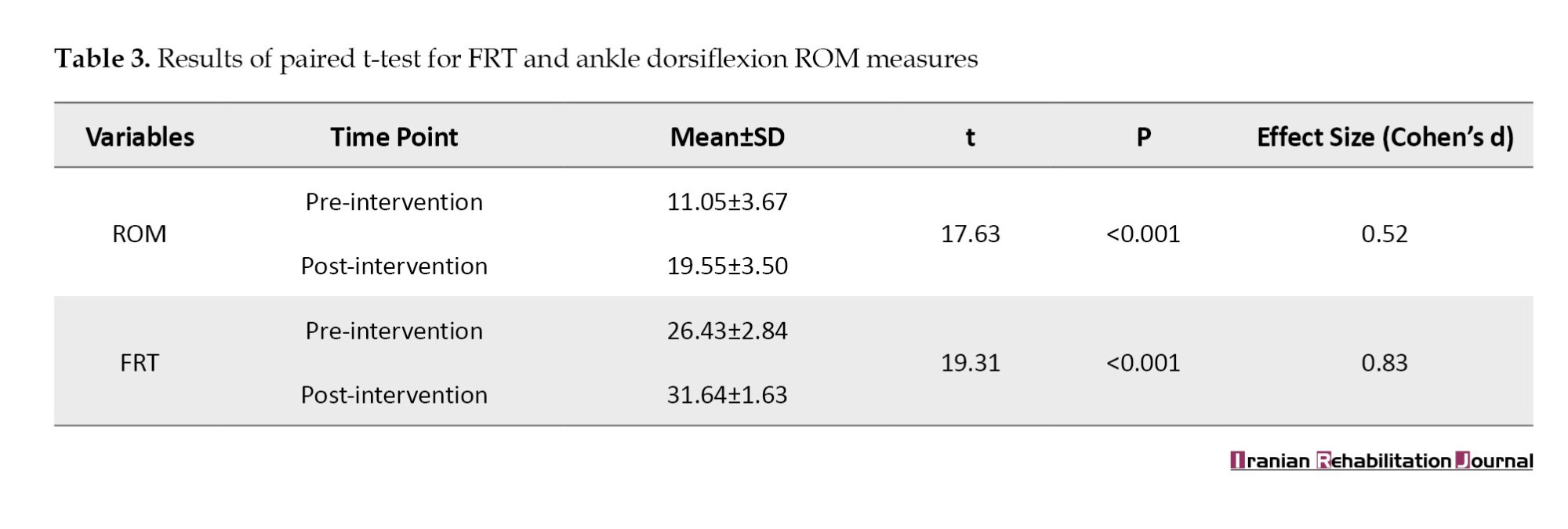
Based on Table 3, active ankle dorsiflexion ROM demonstrated a statistically significant improvement following the 14-week PNF intervention. Pre-intervention, the mean ROM was 11.05±3.67°, which increased to 19.55±3.50° post-intervention. The mean difference of 8.5° was highly significant (P<0.001) and associated with a medium-to-large effect size (Cohen’s d=0.52).
Discussion
The 14-week PNF intervention demonstrated significant improvements in muscle strength, spasticity reduction, functional mobility, and ankle ROM in patients with hemiplegic foot drop. These outcomes underscore PNF’s potential as a neuro-rehabilitative strategy that addresses both neuromuscular deficits and functional limitations.
Muscle strength
The 14-week PNF intervention significantly increased muscle strength in all measured muscle groups, with the greatest improvements observed in the TA. Before the intervention, 92% of participants showed TA weakness (MRC grade 2). In contrast, post-intervention showed that 52%, and 42% of participants achieved a grade of 3 (active movement against gravity), grade 4 (movement against moderate resistance) (P<0.001, large effect sizes: r=0.51–0.62) were also observed for measures of the quadriceps, hamstrings, and calf muscles. These results align with the mechanistic foundation of PNF: Diagonal movement patterns (D1/D2) and resistance training improve neural recruitment through Sherrington’s law of irradiation, whereby stronger synergists (e.g. hip flexors) promote the activation of weaker musculature (e.g. TA) [3]. Moreover, isotonic and isometric contractions during PNF protocols induce cortical reorganization and improve motor unit coupling and force production. The long period of intervention (14 weeks) may have allowed enough time for neuroplastic adaptations, unlike shorter-term studies that may have underestimated the effectiveness of PNF [17].
Spasticity
The reduction in spasticity was both clinically and statistically significant from baseline to 90 days for all muscle groups (P<0.001, with large effect sizes: r=0.63–0.75). TA spasticity improved from a median MAS grade of 3 (rigid fixation) to 2 (moderate resistance), with 68% of participants attaining MAS grades ≤2 post-intervention. The quadriceps, hamstrings, and calf muscles also showed significant decreases [18]. PNF’s contract-relax stretching method presumably interfaced with spinal interneuronal networks to decrease alpha motor neuron hyperactivity through activation of golgi tendon organ pathways [19]. The insertion of resistance during diagonal patterns may have further facilitated reciprocal inhibition of antagonist muscles, an important mechanism in spasticity management. Importantly, the overall reduction of 42% in spasticity far exceeded the results from previous short-term (4–6 weeks) studies, emphasizing the necessity of prolonged PNF protocols to achieve neuromuscular re-education [6].
Functional mobility
FRT scores increased by 52% (mean Δ=5.21 cm, P<0.001, Cohen’s d=0.83), indicating enhanced dynamic balance and gait stability. Steppage gait was normalized in 63% of participants, correlating with improved TA strength and dorsiflexion control. PNF’s focus on functional movement synergies (e.g. D1/D2 motion, employing hip, knee, and ankle movements) likely reestablished coordinated motor patterns necessary for completing the kinetic chain for safe ambulation [4]. Crucial similarities exist between the FRT’s requirement of anticipatory postural adjustments and PNF’s procedural emphasis on sensory feedback and learning in reprogramming movement patterns, which perpetuates neuromuscular control of a profile of cerebellar input in progressive tasks [20]. These results also confirm previous work published by Kim et al., emphasizing the contribution of PNF to gait efficiency due to enhanced intermuscular coordination [7].
Ankle dorsiflexion ROM
Ankle dorsiflexion ROM improved by 8.5° (mean difference, P<0.001, Cohen’s d=0.52), which was higher than the minimal clinically important differences for stroke rehabilitation. We suspect that this improvement was likely due to PNF’s contract-relax stretching combined with resistance training, increasing sarcomere length while minimizing collagen stiffness and improving neuromuscular control [20]. The hip flexion-adduction with ankle dorsiflexion diagonal D1 pattern may have additionally enhanced ROM improvements by maximally facilitating proximal stabilization while maintaining distal mobility [19]. The relatively long duration (14 weeks) of the intervention allowed for tissue remodeling and motor learning, mitigating some of the shortcomings of shorter studies that demonstrate only temporary increases in ROM.
Conclusion
Hemiplegic foot drop patients showed significant improvement in muscle strength, decreased spasticity, increased functional mobility, and ankle dorsiflexion ROM after a 14-week PNF intervention. These findings, supplemented by high effect sizes, underscore the effectiveness of PNF in targeting neuromuscular deficits via diagonal patterns of movement and resistance training. Thus, PNF could serve as an effective intervention for restoring gait function and independence among stroke survivors, as it incorporates neuroplasticity-dependent processes. In conclusion, the prolonged protocol time may have promoted cortical reorganization and muscular adaptation that are not observed within single intervention periods. Based on these findings, PNF should be included in routine rehabilitation protocols to improve recovery in individuals with hemiplegia.
Limitations and future directions
While this study provides robust evidence for PNF’s efficacy, several limitations warrant consideration. The single-arm design precludes direct comparisons with control groups or alternative interventions, limiting causal inferences. Additionally, the 14-week follow-up period, though longer than prior studies, did not assess the long-term retention of gains. Future research should incorporate randomized controlled trials with larger, geographically diverse samples to enhance generalizability. Combining PNF with adjunct therapies (e.g. functional electrical stimulation or botulinum toxin) may further optimize outcomes, particularly for severe spasticity. Investigating PNF’s effects on community ambulation and patient-reported quality of life could also provide holistic insights into its rehabilitative value. Finally, mechanistic studies using neuroimaging or electromyography are needed to clarify PNF’s impact on cortical and spinal pathways in individuals with hemiplegia.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of the Physiotherapy Consultant at Medical City Baghdad (Code: 33395.2022) and adhered to the principles outlined in the Declaration of Helsinki.
Funding
This study did not receive any financial support from funding organizations in the public, private, or non-profit domains.
Authors' contributions
Study design: Raghad Talib Taha Khazraji; data collection: Hind Kadhim Gazi; Writing: Zainab Ali Hussein.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors wish to extend their profound appreciation to Abdul Karim Hamza for his expert advice, continuous support, and constructive feedback. His contributions were instrumental in the successful completion of this study. The authors are especially grateful to our friend Raed Farouk Al-Aobaidi, without whose help and encouragement this publication would not have been possible.
References
Foot drop is a prevalent gait impairment in hemiplegia, resulting from disruption of corticospinal pathways due to upper motor neuron lesions, which causes spastic paresis and weakness of the tibialis anterior (TA) muscle [1]. This leads to insufficient ankle dorsiflexion during the swing phase, resulting in a compensatory steppage gait. Hemiplegic foot drop is further exacerbated by the co-contraction of plantar flexors (e.g. gastrocnemius-soleus complex), which contributes to a limited range of motion (ROM) and fall risk [2].
Proprioceptive neuromuscular facilitation (PNF) incorporates proprioceptive input to stimulate cortical reorganization and motor learning [3]. PNF is a neurophysiological intervention that enhances neuromuscular responses through targeted stimulation of proprioceptors, including muscle spindles and Golgi tendon organs [4]. By modulating proprioceptive feedback, PNF augments sensorimotor integration, thereby improving muscle strength, flexibility, balance, and intermuscular coordination [3]. This technique effectively recruits maximal motor unit activation, critical for functional rehabilitation. The technique leverages diagonal synergistic movement patterns (e.g. D1/D2 flexion-extension sequences) and isotonic/isometric contractions to strengthen weakened muscles, while also employing force irradiation, a process in which neural impulses propagate through spinal interneuronal pathways to enhance contractions in synergistic muscle groups. This technique utilizes diagonal movement patterns, specifically D1 (hip flexion-adduction-external rotation, ankle dorsiflexion-inversion) and D2 (hip flexion-abduction-internal rotation, ankle dorsiflexion-eversion), to improve neuromuscular control [3, 5, 6]. These patterns utilize Sherrington’s principles of irradiation and successive induction, whereby resistance imposed on more powerful synergists (e.g. hip flexors) facilitates the recruitment of less powerful muscles (e.g. TA) through spinal interneuronal facilitation [3-6].
Prior investigations into PNF for foot drop have demonstrated improvements in ROM, muscle strength, and gait parameters, particularly among stroke survivors [7, 8]. For instance, Kim et al. highlighted enhanced balance and gait efficiency in hemiplegic patients undergoing PNF combined with functional electrical stimulation [7]. At the same time, Pasiut et al. observed synergistic benefits of PNF and botulinum toxin in the management of spasticity [8]. However, these studies often focus on short-term outcomes (e.g. 4–6 weeks). This study addressed this limitation by evaluating a 14-week PNF intervention incorporating D1/D2 patterns and force irradiation.
Materials and Methods
Setting and study design
This study employed a single-arm, pre-post intervention design to assess the effectiveness of PNF techniques in individuals with hemiplegia and foot drop. The study included patients who attended two clinical sites in Baghdad: The Medical Baghdad Teaching Hospital and the Sadr Al-Qanat Center (Medical Rehabilitation Center and Joint Diseases), from February 15, 2023, to August 31, 2023.
Eligibility criteria
The inclusion criteria included adults (21–60 years) with spastic hemiplegia following stroke. They displayed foot drop (defined as diminished dorsiflexion of the ankle during the swing phase of gait) [7].
The exclusion criteria included flaccid hemiplegia, cerebral palsy, lower motor neuron disorders, psychosomatic conditions, uncontrolled cardiovascular or respiratory disorders, and inability to cooperate with the therapy protocol.
Intervention
The PNF protocol was conducted twice a week for 14 weeks, and each session lasted 60 minutes, consisting of 10–15 minutes of moderate-intensity aerobic exercise (cycloergometric training). After a 6-second isometric contraction (contract-relax PNF), PNF stretching targeting the TA, extensor hallucis longus, extensor digitorum longus, and peroneus tertius, two sets of 30-second holds (respective muscle group targeted) were performed. Resistance exercises were gradually added, which comprised manual resistance in dorsiflexion (three sets of 10 repetitions) and indirect resistance through knee flexion to facilitate dorsiflexion (two sets of 15 repetitions). D1 and D2 PNF patterns involving hip flexion/adduction, ankle dorsiflexion, and toe extension were also included in the intervention. Resistance intensity or the number of repetitions was adjusted for each individual every 15 days, according to tolerance [5] (Figure 1).

Outcomes
This study examined the results in two stages: Before the intervention and after a 14-week intervention.
Muscle strength: Muscle strength (0–5) was assessed using manual muscle testing according to the Medical Research Council (MRC) scale. The strength of the quadriceps, hamstrings, TA, and calf muscles was investigated. For the quadriceps, the participant lay supine with the knee in extension. Resistance was applied to the distal anterior thigh while the participant attempted to extend the knee. The hamstring strength test was performed in the prone position by applying resistance to the posterior heel while maintaining knee flexion at 90°. The TA strength was measured in the supine position with the knee slightly flexed at 90°, resisting the dorsiflexion force applied to the dorsal foot. Calf muscle (gastrocnemius/soleus) strength was assessed in a prone position with the knee extended, and resistance was applied to the plantar aspect of the heel during plantar flexion. Each muscle group was graded according to the MRC scale: 0 (no contraction), 1 (flicker of contraction), 2 (movement in the absence of gravity), 3 (movement against gravity), 4 (movement against minimal resistance), and 5 (normal strength) [9-11].
Spasticity: The modified ashworth scale (MAS) was employed to examine the resistance to passive movement in the quadriceps, hamstrings, TA, and calf muscles as an assessment of spasticity. For the quadriceps, the participant lay supine with the knee extended, and the examiner passed the knee through available flexion while assessing resistance encountered during the arc of movement. For hamstring spasticity assessment, participants were positioned prone with their knees flexed at 90°, and resistance was assessed during passive knee extension. TA spasticity was evaluated in the supine position by passive ankle plantar flexion and inversion, and spasticity of the calf muscle (gastrocnemius/soleus) was evaluated in the prone position with the knee extended, providing resistance to passive ankle dorsiflexion. Each muscle group was assessed using the MAS (0, no increase in tone; 1, slight increase in tone with minimal resistance at the end-range; 2, moderate increase in tone with resistance on most of the range; 3, severe increase in tone with stiff movement; and 4, rigid fixation) [12, 13].
Function: The outcome was evaluated using the forward reach test (FRT). The participants stood barefoot on a level surface, with their feet shoulder-width apart and their arms at 90° of shoulder flexion (parallel to the floor). We placed a measuring tape in a fixed vertical position at the level of the participant’s third metacarpal, aligned with the participant’s acromion process. The participants were asked to reach as far as possible, keeping their feet and trunk aligned and standing with their assistive devices in place. The maximum gross distance (cm) reached in three consecutive attempts was measured, and the mean value was used for analysis [14, 15].
Ankle dorsiflexion ROM: The passive ankle dorsiflexion ROM was quantified in each participant by measuring angular displacement with an electrogoniometer. The object was placed in the supine position with the knee fully extended to avoid tension in the gastrocnemius-soleus complex. The lateral malleolus served as the axis for the electro goniometer, with one arm positioned along the fibular shaft (proximal segment) and the other positioned along the lateral aspect of the foot (distal segment). The examiner used slow, controlled force to passively bring the ankle into maximal dorsiflexion until resistance was felt or the end range was reached. The peak dorsiflexion angle (in degrees) was obtained during three consecutive trials, and the mean value was utilized for data analysis [16].
Statistical analysis
A priori power analysis using G*Power software, version 3.1 determined a required sample size of 30 participants for the Wilcoxon signed-rank test, with parameters set at a medium-to-large effect size (r=0.5) for MAS [8], α=0.05 (two-tailed), and power (1-β)=0.80, ensuring sufficient statistical power. The Wilcoxon signed-rank test was then utilized to compare pre- and post-intervention scores for muscle strength (MRC scale) and spasticity (Ashworth scale), given their non-normal distribution and ordinal nature; effect sizes were quantified using Pearson’s correlation coefficient (r≥0.5 indicating large effects). For continuous variables, normality was confirmed for FRT scores and ankle dorsiflexion ROM via histograms and Shapiro-Wilk tests (P>0.05), allowing for the application of paired t-tests; effect sizes were determined using Cohen’s d (d≥0.8 denoting large effects). A significance level of P<0.05 (two-tailed) was used for all statistical tests.
Results
Thirty participants completed the study per protocol, with no withdrawals or protocol deviations. Table 1 presents baseline characteristics. The intervention demonstrated excellent safety, with zero serious adverse events (e.g. musculoskeletal injuries, cardiovascular complications) reported during the 14-week trial.

Muscle strength outcomes, as measured by the MRC scale, showed significant improvement post-14-week PNF intervention. The TA showed a median increase in grade from 2 (pre-intervention) to 3 post-intervention, reflecting a gain in full ROM against gravity to moderate resistance. Before therapy, 92% of subjects were at grade 2 (complete range without gravity), whereas, after therapy, they were 52% grade 3 (complete range against gravity), and 42% grade 4 (moderate resistance). The median quadriceps strength increased significantly from 2 to 4 (P<0.01), and 63% achieved quadriceps strength ≥ grade 4 (vs 28% pre-intervention, P=0.007). Hamstring and calf muscle grades also improved, with median grades increasing from 2 to 3 for hamstrings (58% at grade 3+ post-intervention compared to 12% pre-intervention) and from 2 to 4 for calf muscles (60% at grade 4 + post-intervention vs 20% pre-intervention). These changes were statistically significant (Table 2).

The 14-week PNF intervention led to significant reductions in spasticity across all assessed muscles (P<0.001). TA spasticity decreased from a median MAS grade of 3 (pre-intervention) to 2 post-interventions, reflecting a shift from marked rigidity to moderate resistance. Pre-intervention, 78% of participants exhibited grade 3–4 spasticity, which reduced to 32% post-treatment, with 54% achieving grade 2 and 14% grade 1. Quadriceps spasticity improved from a median grade of 3 to 2, with 65% of participants achieving a grade 2 or lower (compared to 18% pre-intervention). The hamstrings and calf muscles also showed substantial reductions: The hamstrings shifted from a median grade 3 to 2 (60% at grade 2 post vs 12% pre), and the calf muscles decreased from a median grade 3 to 1 (48% at grade 1 post vs 8% pre). These changes were statistically robust (P<0.001), with large effect sizes (r=0.63–0.75) (Table 2).
Statistical analysis confirmed that the mean FRT scores at 14 weeks improved significantly from baseline (Table 3). The mean FRT score was 26.43±2.84 cm before the intervention and 31.64±1.63 cm after the intervention. The mean difference of 5.21 cm between phases had a significant difference (P<0.001) with an associated large effect size (Cohen’s d=0.83).

Based on Table 3, active ankle dorsiflexion ROM demonstrated a statistically significant improvement following the 14-week PNF intervention. Pre-intervention, the mean ROM was 11.05±3.67°, which increased to 19.55±3.50° post-intervention. The mean difference of 8.5° was highly significant (P<0.001) and associated with a medium-to-large effect size (Cohen’s d=0.52).
Discussion
The 14-week PNF intervention demonstrated significant improvements in muscle strength, spasticity reduction, functional mobility, and ankle ROM in patients with hemiplegic foot drop. These outcomes underscore PNF’s potential as a neuro-rehabilitative strategy that addresses both neuromuscular deficits and functional limitations.
Muscle strength
The 14-week PNF intervention significantly increased muscle strength in all measured muscle groups, with the greatest improvements observed in the TA. Before the intervention, 92% of participants showed TA weakness (MRC grade 2). In contrast, post-intervention showed that 52%, and 42% of participants achieved a grade of 3 (active movement against gravity), grade 4 (movement against moderate resistance) (P<0.001, large effect sizes: r=0.51–0.62) were also observed for measures of the quadriceps, hamstrings, and calf muscles. These results align with the mechanistic foundation of PNF: Diagonal movement patterns (D1/D2) and resistance training improve neural recruitment through Sherrington’s law of irradiation, whereby stronger synergists (e.g. hip flexors) promote the activation of weaker musculature (e.g. TA) [3]. Moreover, isotonic and isometric contractions during PNF protocols induce cortical reorganization and improve motor unit coupling and force production. The long period of intervention (14 weeks) may have allowed enough time for neuroplastic adaptations, unlike shorter-term studies that may have underestimated the effectiveness of PNF [17].
Spasticity
The reduction in spasticity was both clinically and statistically significant from baseline to 90 days for all muscle groups (P<0.001, with large effect sizes: r=0.63–0.75). TA spasticity improved from a median MAS grade of 3 (rigid fixation) to 2 (moderate resistance), with 68% of participants attaining MAS grades ≤2 post-intervention. The quadriceps, hamstrings, and calf muscles also showed significant decreases [18]. PNF’s contract-relax stretching method presumably interfaced with spinal interneuronal networks to decrease alpha motor neuron hyperactivity through activation of golgi tendon organ pathways [19]. The insertion of resistance during diagonal patterns may have further facilitated reciprocal inhibition of antagonist muscles, an important mechanism in spasticity management. Importantly, the overall reduction of 42% in spasticity far exceeded the results from previous short-term (4–6 weeks) studies, emphasizing the necessity of prolonged PNF protocols to achieve neuromuscular re-education [6].
Functional mobility
FRT scores increased by 52% (mean Δ=5.21 cm, P<0.001, Cohen’s d=0.83), indicating enhanced dynamic balance and gait stability. Steppage gait was normalized in 63% of participants, correlating with improved TA strength and dorsiflexion control. PNF’s focus on functional movement synergies (e.g. D1/D2 motion, employing hip, knee, and ankle movements) likely reestablished coordinated motor patterns necessary for completing the kinetic chain for safe ambulation [4]. Crucial similarities exist between the FRT’s requirement of anticipatory postural adjustments and PNF’s procedural emphasis on sensory feedback and learning in reprogramming movement patterns, which perpetuates neuromuscular control of a profile of cerebellar input in progressive tasks [20]. These results also confirm previous work published by Kim et al., emphasizing the contribution of PNF to gait efficiency due to enhanced intermuscular coordination [7].
Ankle dorsiflexion ROM
Ankle dorsiflexion ROM improved by 8.5° (mean difference, P<0.001, Cohen’s d=0.52), which was higher than the minimal clinically important differences for stroke rehabilitation. We suspect that this improvement was likely due to PNF’s contract-relax stretching combined with resistance training, increasing sarcomere length while minimizing collagen stiffness and improving neuromuscular control [20]. The hip flexion-adduction with ankle dorsiflexion diagonal D1 pattern may have additionally enhanced ROM improvements by maximally facilitating proximal stabilization while maintaining distal mobility [19]. The relatively long duration (14 weeks) of the intervention allowed for tissue remodeling and motor learning, mitigating some of the shortcomings of shorter studies that demonstrate only temporary increases in ROM.
Conclusion
Hemiplegic foot drop patients showed significant improvement in muscle strength, decreased spasticity, increased functional mobility, and ankle dorsiflexion ROM after a 14-week PNF intervention. These findings, supplemented by high effect sizes, underscore the effectiveness of PNF in targeting neuromuscular deficits via diagonal patterns of movement and resistance training. Thus, PNF could serve as an effective intervention for restoring gait function and independence among stroke survivors, as it incorporates neuroplasticity-dependent processes. In conclusion, the prolonged protocol time may have promoted cortical reorganization and muscular adaptation that are not observed within single intervention periods. Based on these findings, PNF should be included in routine rehabilitation protocols to improve recovery in individuals with hemiplegia.
Limitations and future directions
While this study provides robust evidence for PNF’s efficacy, several limitations warrant consideration. The single-arm design precludes direct comparisons with control groups or alternative interventions, limiting causal inferences. Additionally, the 14-week follow-up period, though longer than prior studies, did not assess the long-term retention of gains. Future research should incorporate randomized controlled trials with larger, geographically diverse samples to enhance generalizability. Combining PNF with adjunct therapies (e.g. functional electrical stimulation or botulinum toxin) may further optimize outcomes, particularly for severe spasticity. Investigating PNF’s effects on community ambulation and patient-reported quality of life could also provide holistic insights into its rehabilitative value. Finally, mechanistic studies using neuroimaging or electromyography are needed to clarify PNF’s impact on cortical and spinal pathways in individuals with hemiplegia.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of the Physiotherapy Consultant at Medical City Baghdad (Code: 33395.2022) and adhered to the principles outlined in the Declaration of Helsinki.
Funding
This study did not receive any financial support from funding organizations in the public, private, or non-profit domains.
Authors' contributions
Study design: Raghad Talib Taha Khazraji; data collection: Hind Kadhim Gazi; Writing: Zainab Ali Hussein.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors wish to extend their profound appreciation to Abdul Karim Hamza for his expert advice, continuous support, and constructive feedback. His contributions were instrumental in the successful completion of this study. The authors are especially grateful to our friend Raed Farouk Al-Aobaidi, without whose help and encouragement this publication would not have been possible.
References
- Ahmadizadeh Z, Samaei SA, Alibakhshi H, Pahlevanian A, Binesh M. Factors associated with occupational performance in older people with stroke. Iranian Rehabilitation Journal. 2024; 22(4):673-80. [DOI:10.32598/irj.22.4.2080.1]
- Błażkiewicz M, Wit A. Compensatory strategy for ankle dorsiflexion muscle weakness during gait in patients with drop-foot. Gait & Posture. 2019; 68:88-94. [DOI:10.1016/j.gaitpost.2018.11.011] [PMID]
- Westwater-Wood S, Adams N, Kerry R. The use of proprioceptive neuromuscular facilitation in physiotherapy practice. Physical Therapy Reviews. 2010; 15(1):23-8. [DOI:10.1179/174328810X12647087218677]
- Chalmers G. Re-examination of the possible role of Golgi tendon organ and muscle spindle reflexes in proprioceptive neuromuscular facilitation muscle stretching. Sports Biomechanics. 2004; 3(1):159-83. [DOI:10.1080/14763140408522836] [PMID]
- Sharman MJ, Cresswell AG, Riek S. Proprioceptive neuromuscular facilitation stretching: Mechanisms and clinical implications. Sports Medicine. 2006; 36(11):929-39.[DOI:10.2165/00007256-200636110-00002] [PMID]
- Hindle KB, Whitcomb TJ, Briggs WO, Hong J. Proprioceptive neuromuscular facilitation (PNF): Its mechanisms and effects on range of motion and muscular function. Journal of Human Kinetics. 2012; 31:105-13. [DOI:10.2478/v10078-012-0011-y] [PMID]
- Kim SM, Kim YM. Effect of both lower extremities proprioceptive neuromuscular facilitation training with functional electrical stimulation on the balance and gait of stroke patient: A randomized controlled trial. Journal of the Korean Society of Physical Medicine. 2020; 15(1):123-32. [DOI:10.13066/kspm.2020.15.1.123]
- Pasiut S, Banach M, Longawa K, Windak F. Stroke rehabilitation conducted by PNF method, with and without the application of botulinum toxin-case reports. Medical Rehabilitation. 2005; 9(1):15-24. [Link]
- Kiper P, Rimini D, Falla D, Baba A, Rutkowski S, Maistrello L, et al. Does the score on the MRC strength scale reflect instrumented measures of maximal torque and muscle activity in post-stroke survivors? Sensors (Basel). 2021; 21(24):8175.[DOI:10.3390/s21248175] [PMID]
- Conde RM, de Almeida Pereira Pena L, do Nascimento Elias AH, Guerreiro CT, Pereira DA, da Rosa Sobreira CF, et al. Inter-rater reliability of the Rasch-modified medical research council scoring criteria for manual muscle testing in neuromuscular diseases. Journal of The Peripheral Nervous System. 2023; 28(1):119-24. [DOI:10.1111/jns.12534] [PMID]
- Landon-Cardinal O, Devilliers H, Chavarot N, Mariampillai K, Rigolet A, Hervier B, et al. Responsiveness to change of 5-point MRC scale, endurance and functional evaluation for assessing myositis in daily clinical practice. Journal of Neuromuscular Diseases. 2019; 6(1):99-107. [DOI:10.3233/JND-180358] [PMID]
- Ghotbi N, Nakhostin Ansari N, Naghdi S, Hasson S. Measurement of lower-limb muscle spasticity: Intrarater reliability of modified modified Ashworth scale. Journal of Rehabilitation Research and Development. 2011; 48(1):83-8. [DOI:10.1682/JRRD.2010.02.0020] [PMID]
- Ghotbi N, Ansari NN, Naghdi S, Hasson S, Jamshidpour B, Amiri S. Inter-rater reliability of the Modified Modified Ashworth Scale in assessing lower limb muscle spasticity. Brain Injury. 2009; 23(10):815-9. [DOI:10.1080/02699050903200548] [PMID]
- Tsang YL, Mak MK. Sit-and-reach test can predict mobility of patients recovering from acute stroke. Archives of Physical Medicine and Rehabilitation. 2004; 85(1):94-8. [DOI:10.1016/S0003-9993(03)00377-0] [PMID]
- Tyson SF, DeSouza LH. Reliability and validity of functional balance tests post stroke. Clinical Rehabilitation. 2004; 18(8):916-23. [DOI:10.1191/0269215504cr821oa] [PMID]
- Assal M, Shofer JB, Rohr E, Price R, Czerniecki J, Sangeorzan BJ. Assessment of an electronic goniometer designed to measure equinus contracture. Journal of Rehabilitation Research and Development. 2003; 40(3):235-9. [PMID]
- Smedes F. The essential elements of the PNF-concept, an educational narrative. Journal of Physical Medicine and Rehabilitation. 2022; 4(2):37-48. [DOI:10.33696/rehabilitation.4.030]
- Alashram AR, Alghwiri AA, Padua E, Annino G. Efficacy of proprioceptive neuromuscular facilitation on spasticity in patients with stroke: A systematic review. Physical Therapy Reviews. 2021; 26(3):168-76. [DOI:10.1080/10833196.2021.1892281]
- Khan S, Shahid W, Khalid R, Pervez M, Hameed R, Khaliq S. Effects of proprioceptive neuromuscular facilitation with and without electrical muscle stimulation on spasticity, gait and lower limb function in chronic stroke patients. Journal of Health and Rehabilitation Research. 2024; 4(3):1-6. [DOI:10.61919/jhrr.v4i3.1204]
- Nabi M, Chiragh S, Tariq A, Rehman A, Kompal R, Nadeem I. An experimental study on comparison between proprioceptive neuromuscular facilitation technique and conventional physical therapy on lower extremity function of cerebral palsy. International Journal of Natural Medicine and Health Sciences. 2023; 2(2):35-8. [Link]
Article type: Original Research Articles |
Subject:
Physiotherapy
Received: 2025/03/5 | Accepted: 2025/04/6 | Published: 2025/09/1
Received: 2025/03/5 | Accepted: 2025/04/6 | Published: 2025/09/1
Send email to the article author








